
Six batches of NovoPen Echo insulin cartridge holders have been recalled.
Subscribe now for unlimited access.
$0/
(min cost $0)
or signup to continue reading
Authorities believe there is a risk that they may crack or break if exposed to chemicals in certain cleaning agents.
People who use a cracked or broken cartridge holder could be delievered a smaller dose of insulin than expected leading to high blood sugar levels, potentially putting the person at risk of hyperglycemia.
Novo Nordisk has advised that if people clean the pens as described in the User Guide, there is no reason that cracking of the cartridge holder will occur.
The batches involved are:
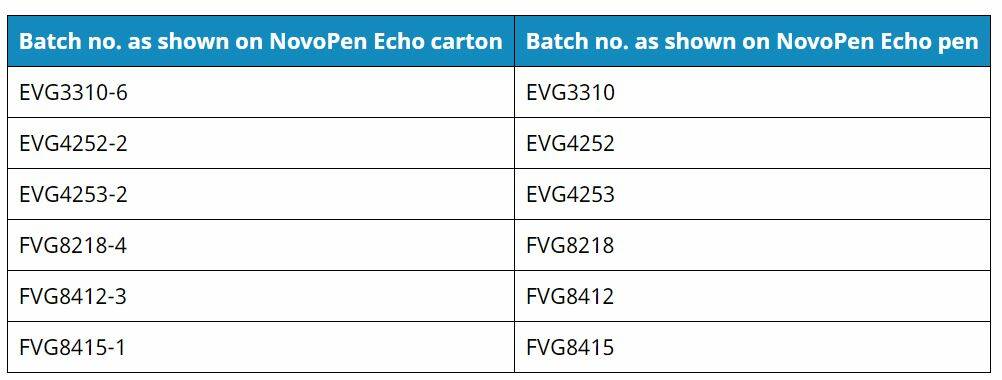
The recall only relates to some batches of the cartridge holders and there are no reported problems with the insulin being administered.
People with diabetes who use a NovoPen Echo should immediately check the batch number.
What to do if you are using an affected NovoPen Echo
- Do not stop treatment without consulting your doctor.
- Register your contact details (name, address, phone number, email and number of affected cartridge holders) either via www.novonordisk.com.au or at www.novonordisk.com/novopenecho5.html in order to receive a replacement cartridge holder. A replacement should arrive in about seven days.
- You should measure your blood sugar levels as instructed by your health care provider and more frequently if symptoms of too high or too low blood sugar levels develop unexpectedly.
- In the event that you experience symptoms of too high blood sugar levels involving this product, contact your doctor for advice.
- Report any adverse events or complaints to the NovoCare Customer Care Centre, which can be reached at 0800 733 737 or via email at aunrccc@novonordisk.com.

